 Only the bonding molecular orbitals participate in the bond formation. Antibonding orbitals have more energy compared to the bonding molecular orbitals. Thus thereby avoiding the extra electron interacting with other electrons. When there is a presence of extra electron like in the case of H - 2 the extra electron will be present in the antibonding orbital. The Following Figure Shows the Formation of Antibonding Orbitals. NCERT notes Class 11 Chemistry Chapter 12 Organic chemistry- some basic principles and techniques.NCERT Exemplar Class 11 Chemistry Solutions Chapter 12 Organic chemistry- some basic principles and techniques. 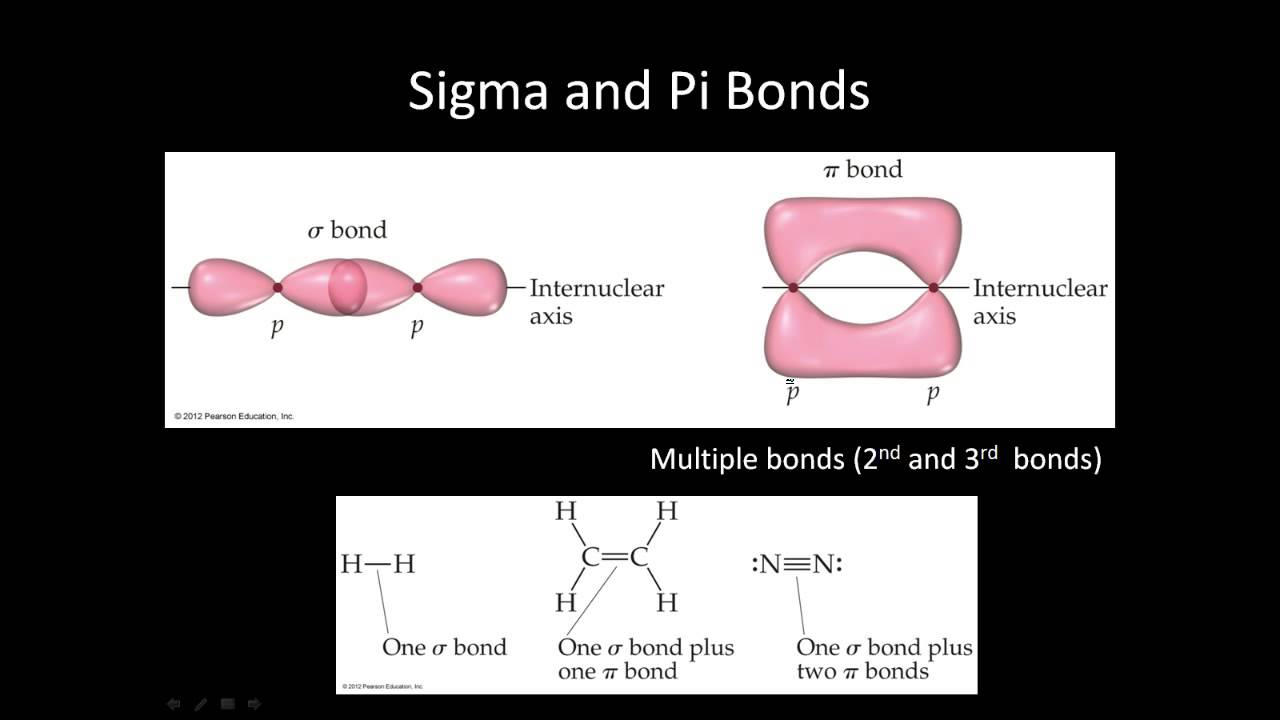
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
